Current Projects
Funded by the NIH, Department of Defense and other agencies, our research has been focused on the following areas.
Airway innate immunity in host defense against pathogens
We are studying how airway epithelial cells respond to respiratory bacteria (e.g. non-typeable Heamophilus influenzae) and viruses (rhinovirus, influenza virus and SARS-COV-2). Some of the innate molecules we have studied include TLR2, BPIFA1 (SPLUNC1), IRAK-M and Tollip. We are particularly interested in the role of negative immune regulators in the resolution of infection and inflammation, and how asthma and COPD conditions (e.g. allergic inflammation and smoking) modulate the expression and functions of negative immune regulators.
Genetic variation in innate immunity and asthma
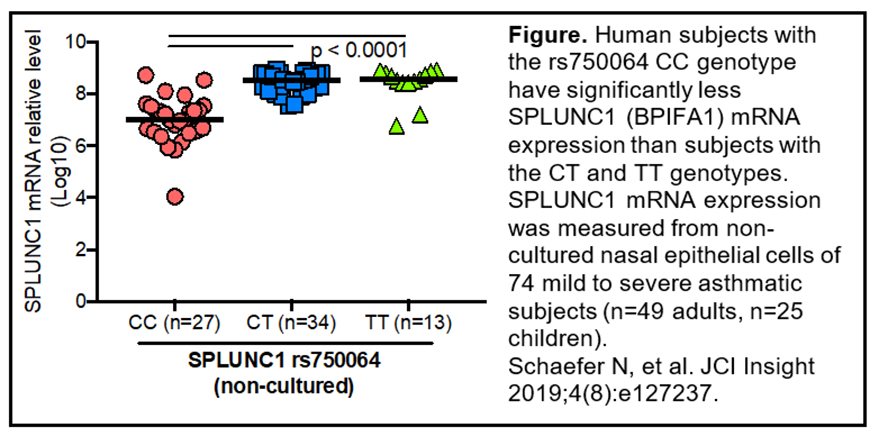
Genetic factors contribute to the host response to environmental factors such as infection, allergens and tobacco or electronic cigarette smoke exposure. We have found that single nucleotide polymorphisms (SNPs) of BPIFA1 (e.g. the rs750064 CC genotype, see the figure) and Tollip contribute to the gene expression levels, airway obstruction and serum IgE levels in asthma, and airway epithelial cell responses to viral infection and type 2 cytokine IL-13. We continue to study the function and underlying mechanisms of genetic variation in host susceptibility to infections and other environmental factors.
Mitochondrial dysfunction in airway disease
Infection, inflammation and other pathological conditions affect mitochondrial biogenesis and functions. We study how different physiological and pathological factors regulate mitochondrial functions in the context of obesity, asthma and infection.
Impact of tobacco smoke and electronic cigarettes on lung immunity
Tobacco smoke and electronic cigarette use remains a significant healthcare challenge. We use animal models, cell cultures and precision-cut lung slice system to study how tobacco smoke and electronic cigarette exposures impair proximal and distal airway epithelial and lung parenchymal responses to bacterial and viral infections.
